Medical Device Validation process Consultant
Ensure Compliance and Product Safety with Robust Validation Documentation
At Operon Strategist, we provide comprehensive validation documentation services tailored for medical product manufacturers in the Netherlands. Our expertise ensures that your processes, equipment, and facilities meet the rigorous requirements of EU MDR, ISO standards, and other global regulatory frameworks.
What is medical device Validation?
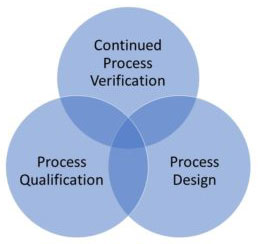
Medical Device Process Validation is a process of establishing documentary evidence demonstrating that a procedure, process or activity carried out in production maintains the desired level of compliance at all stages. In straightforward words, the interaction approval for a medical device is the assortment and assessment of information, from the cycle configuration stage till the creation, as it gives logical proof that the cycle is able to do reliably giving quality items. In simple words, the process validation medical device is the collection and evaluation of data, from the process design stage till the production, as it gives scientific evidence that the process is capable of consistently providing quality products.
What is the difference between process verification and validation?
In medical device manufacturing, understanding the distinction between process verification and process validation is crucial for maintaining regulatory compliance and product quality.
Process verification involves checking individual products to ensure they meet specified requirements, typically through inspection or testing. While this method is effective for small batches, it becomes impractical and costly in large-scale production.
This is where process validation becomes essential. According to the US FDA, process validation establishes documented evidence that a manufacturing process consistently produces products that meet predetermined specifications. Rather than testing each device, validation ensures the process itself is reliable, efficient, and repeatable.
As an expert ISO 13485 medical device consultant in the Netherlands, Operon Strategist helps manufacturers implement both verification and validation processes in line with regulatory expectations such as EU MDR, US FDA, and local Dutch guidelines. Our team ensures that your quality systems are robust, compliant, and tailored for successful market access.
Get Expert Help with Your Medical Device Validation
Fill the Form or Mail Us to: enquiry@operonstrategist.com
Medical Device Process Validation is divided into the following sub-sections:
- HVAC validation
- Equipment validation
- Process validation
- Facilities validation
- Cleaning validation
- Analytical method validation
- Personnel validation
- Packaging validation
- Computer system validation
Why choose Operon Strategist as validation process consultant?
- Proven Track Record in the Netherlands and EU: We’ve helped several Dutch and EU-based manufacturers achieve successful validation outcomes.
- Regulatory Expertise: Our consultants stay updated with evolving EU MDR and ISO regulations, ensuring your documentation meets the latest expectations.
- Turnkey Project Capability: We offer end-to-end support, including validation planning, protocol drafting, execution, report writing, and audit preparation.
- Quality with Speed: We understand the pressures of product launch timelines. Our process is efficient without compromising on quality or compliance.
Let the Experts Handle Your Process Validation – Connect with Operon Strategist Now!
FAQs
1. What is validation documentation in medical product manufacturing?
Validation documentation refers to a structured set of records that demonstrate a medical device manufacturing process meets regulatory standards. It includes process validation, equipment validation, software validation, cleaning validation, and sterilization validation.
2. Why is validation documentation important for medical devices?
Validation documentation ensures that all processes in medical device manufacturing are consistent, reproducible, and compliant with regulatory standards such as ISO 13485, FDA 21 CFR Part 820, and EU MDR. It helps prevent defects, enhances product safety, and supports regulatory approvals.
3. What are the key types of validation required in medical device manufacturing?
The key validation types include process validation, equipment validation, software validation, cleaning validation, and sterilization validation. Each ensures specific aspects of manufacturing meet regulatory and quality requirements.
4. How does Operon Strategist help with validation documentation?
Operon Strategist provides end-to-end validation documentation services, including creating validation master plans, protocols, reports, and execution support. We ensure compliance with international regulatory standards and help streamline the approval process.
5. Which regulatory standards require validation documentation?
Validation documentation is required for compliance with various regulatory standards, including ISO 13485, FDA 21 CFR Part 820, EU MDR, CE Marking, UKCA, and MDSAP.
