Clean Room Design Consultant for Medical Devices
Ensure Regulatory-Ready Clean Rooms for Your Medical Device Facility
At Operon Strategist, we help medical device manufacturers in the Netherlands establish and validate clean rooms that meet international standards, including ISO 14644 and EU GMP requirements. Whether you’re a startup or an established company expanding operations, our expert clean room consulting services are designed to ensure compliance, optimize production efficiency, and support your regulatory approvals.

What is a clean room?
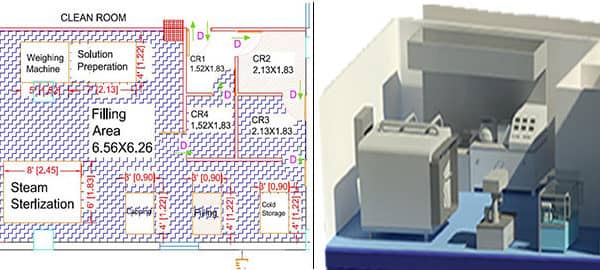
Cleanrooms are the enclosed area within manufacturing facility, these are specially designed rooms to control air pollutant level, humidity and personal access to meet environmental conditions. Clean Room helps in establishing & maintaining an environment with a low level of environmental pollutants such as dust, airborne microbes, aerosol particles & chemical vapors. For that, clean room design is under the expertise is more important. Typically, Manufacturers/producers of medical devices, pharmaceuticals, and biotechnology products need clean rooms for their units.
Why Clean Rooms Matter in Medical Device Manufacturing
Clean rooms are controlled environments where airborne particles, temperature, humidity, and microbial contamination are monitored and maintained within strict limits. For medical devices, especially those classified under higher risk categories, maintaining product sterility and minimizing contamination is not just a quality measure—it’s a regulatory requirement.
Clean room compliance is essential for:
- CE Marking under EU MDR
- ISO 13485 Certification
- Notified Body Audits
- FDA and MDSAP inspections
Get Expert Help with Your Medical Device Validation
Fill the Form or Mail Us to: enquiry@operonstrategist.com
Benefits of Working with Operon Strategist
- Over 15 years of regulatory and facility design expertise
- Localized support tailored for Dutch regulations and EU MDR
- Multi-disciplinary team: engineers, QA/RA experts, and project managers
- Strong understanding of ISO 14644, ISO 13485, and EU GMP
- Proven track record with turnkey clean room projects
Start Your Clean Room Project in the Netherlands with Confidence
Building a compliant clean room is more than just construction it requires strategic planning, regulatory compliance, and seamless execution. At Operon Strategist, we help you design and implement clean rooms that meet EU GMP, ISO 14644, and ISO 13485 standards making your medical device manufacturing facility inspection-ready from day one.
Ready to set up your clean room in the Netherlands?
FAQs
1. What is clean room guidance in medical product manufacturing?
Clean room guidance involves designing and maintaining a controlled environment with minimal contamination levels to ensure compliance with regulatory standards for medical product manufacturing.
2. Why is clean room compliance important for medical device manufacturing?
Clean room compliance ensures product safety by preventing contamination, meeting international regulatory requirements such as ISO 14644, and improving product quality.
3. What are the different clean room classifications?
Clean rooms are classified based on particle count per cubic meter, as per ISO 14644-1, ranging from ISO Class 1 (most stringent) to ISO Class 9 (least stringent).
4. How does Operon Strategist help with clean room design and compliance?
Operon Strategist provides end-to-end clean room consulting, including design layout, classification recommendations, validation services, regulatory compliance assessment, and operational best practices for contamination control.
5. What industries require clean room consulting services?
Industries such as medical device manufacturing, pharmaceuticals, biotechnology, and healthcare product manufacturing require clean rooms for regulatory compliance and product safety.
