Medical Device Industry in Saudi Arabia
Saudi Arabia, the largest and most populous country in the Gulf Cooperation Council (GCC), has a population exceeding 37 million and a GDP of over $1.1 trillion. The demand for healthcare services and medical devices is rising, driven by several key factors:
- A growing and aging population
- Increasing public awareness of health and wellness
- Government initiatives to enhance healthcare quality and accessibility
Register a Medical Device in Saudi Arabia
As a result, Saudi Arabia’s medical device market is experiencing significant growth, projected to expand from $2.0 billion in 2022 to $3.3 billion by 2032, at a CAGR of 5.9%.
However, manufacturers must navigate the Saudi Food and Drug Authority (SFDA) approval process before entering this lucrative market. Let’s explore how you can get your medical device registered and start selling in Saudi Arabia.
This guide provides a step-by-step process for registering a medical device in Saudi Arabia. It highlights the role of Operon Strategist, a trusted regulatory consulting firm, in facilitating compliance.
Process to Register a Medical Device in Saudi Arabia
The Saudi Food and Drug Authority (SFDA) has introduced a new registration pathway requiring a Technical File Assessment (TFA) for market authorization. The SFDA has also implemented its own classification system for medical devices. Unlike many other markets, prior approval in the country of origin is not a prerequisite for registration in Saudi Arabia. Additionally, reference country approval is not mandatory for Medical Device Marketing Authorization (MDMA) registration. However, any existing approvals from other markets may be requested as supporting documentation during the registration process.
Regulatory Framework for Medical Device Registration in Saudi Arabia
The SFDA regulates medical devices through the Medical Devices Interim Regulation, which aligns with international standards such as those from the International Medical Device Regulators Forum (IMDRF) and Global Harmonization Task Force (GHTF). Registration is mandatory for all medical devices before they can be marketed in Saudi Arabia.
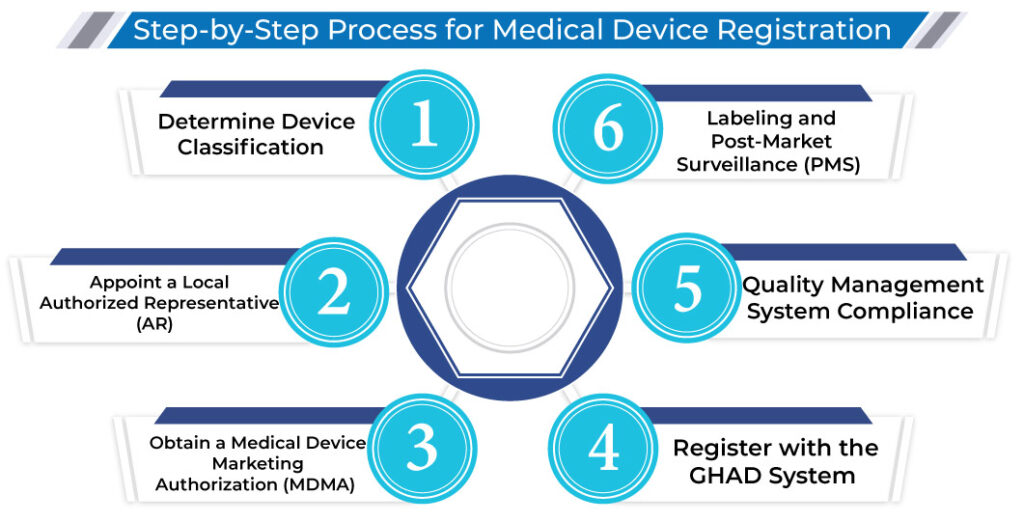
Step-by-Step Process to Register a Medical Device in Saudi Arabia
- Determine Device Classification
Medical devices are classified based on risk, following the IMDRF classification system:
- Class A (Low risk)
- Class B (Low to moderate risk)
- Class C (Moderate to high risk)
- Class D (High risk)
The classification determines the level of scrutiny required during the registration process.
In Vitro Diagnostic (IVD) Device Classification: IVD medical devices are classified based on their intended use and associated risk:
- Class A – Low individual and public health risk (e.g., general laboratory equipment)
- Class B – Moderate individual risk and low public health risk (e.g., pregnancy tests)
- Class C – High individual risk and/or moderate public health risk (e.g., blood glucose monitoring systems)
- Class D – High individual and public health risk (e.g., HIV or hepatitis diagnostic tests)
- Appoint a Local Authorized Representative (AR)
International manufacturers must appoint a Saudi-based Authorized Representative (AR) to liaise with SFDA. The AR is responsible for submitting applications, handling communications, and ensuring compliance with local regulations.
- Obtain a Medical Device Marketing Authorization (MDMA)
MDMA is a crucial approval issued by SFDA, which ensures the device meets safety and performance requirements. The process includes:
- Submitting a Technical File with details on design, risk management, and quality assurance
- Providing evidence of compliance with international standards (ISO 13485, CE marking, FDA approval, etc.)
- Demonstrating clinical data and safety information for high-risk devices
- Register with the GHAD System
The SFDA uses an online portal called GHAD for medical device registration. The manufacturer or AR must create an account and submit the required documentation electronically.
Manufacturers must comply with ISO 13485:2016, an international standard for quality management systems specific to medical devices. SFDA may conduct audits to ensure compliance.
- Labeling and Post-Market Surveillance (PMS)
All medical devices must have labeling in Arabic and English, including essential safety and usage information. Post-market surveillance, including reporting adverse events and recalls, is mandatory. HAD
Get Expert Regulatory Consulting Services to Register a Medical Device in Saudi Arabia
Role of Operon Strategist in Medical Device Registration
Navigating SFDA regulations can be complex, and non-compliance can lead to delays or rejections. Operon Strategist, a leading regulatory consulting firm, plays a critical role in ensuring a smooth registration process. Their expertise includes:
- Device Classification Assistance: Ensuring correct classification to avoid regulatory hurdles.
- Technical File Preparation: Assisting in compiling comprehensive documentation for SFDA submission.
- Local Representation Support: Connecting manufacturers with reliable Authorized Representatives.
- Ghad System Registration: Streamlining the online application process.
- Quality Compliance Consultation: Ensuring adherence to ISO 13485 and other regulatory standards.
- Post-Market Compliance Management: Helping with surveillance, recalls, and reporting requirements.
🔹 Our Key Services:
- SFDA Registration Assistance – Helping manufacturers navigate SFDA product registration.
- SFDA Authorized Representative Service Assistance – Providing AR services for seamless regulatory compliance.
- Quality Management System (QMS) Compliance – Ensuring adherence to ISO 13485 and SFDA standards.
- Technical File Preparation – Assisting in compiling SFDA-compliant technical documentation.
- Turnkey Medical Device Project Solutions – From facility setup to regulatory approvals.
- Regulatory Training & Support – Educating manufacturers on evolving compliance requirements.




