Medical Device Manufacturing Facility Setup in Costa Rica
Strategic Manufacturing Plant Layout Design for Medical Devices in Costa Rica
The foundation of a successful medical device manufacturing facility lies in a layout that balances operational excellence with unwavering regulatory compliance. In Costa Rica’s rapidly growing medical hub, manufacturers must adhere to stringent global standards—including the US FDA, CDSCO, SFDA, and CE Marking—to ensure product safety and quality.
Whether you are producing orthopedic implants, disposables, primary packaging, or pharmaceutical products, your facility must be meticulously designed to meet cGMP (Current Good Manufacturing Practice) and international regulatory mandates.
Comprehensive Facility Setup & Digital Integration
At Operon Strategist, we provide end-to-end solutions specifically tailored for the Costa Rican medical device sector. Our services bridge the gap between initial planning and a fully operational, audit-ready facility.
Streamlined Manufacturing with MES (Manufacturing Execution System)
To stay competitive, modern facilities must embrace digital transformation. We integrate advanced MES solutions into your manufacturing process. This allows for real-time production monitoring, enhanced operational efficiency, and total product traceability—ensuring your quality control is data-driven and airtight.
Let's Grow Your Business Together
Let’s have word about your next project.
The Blueprint for Operational Excellence
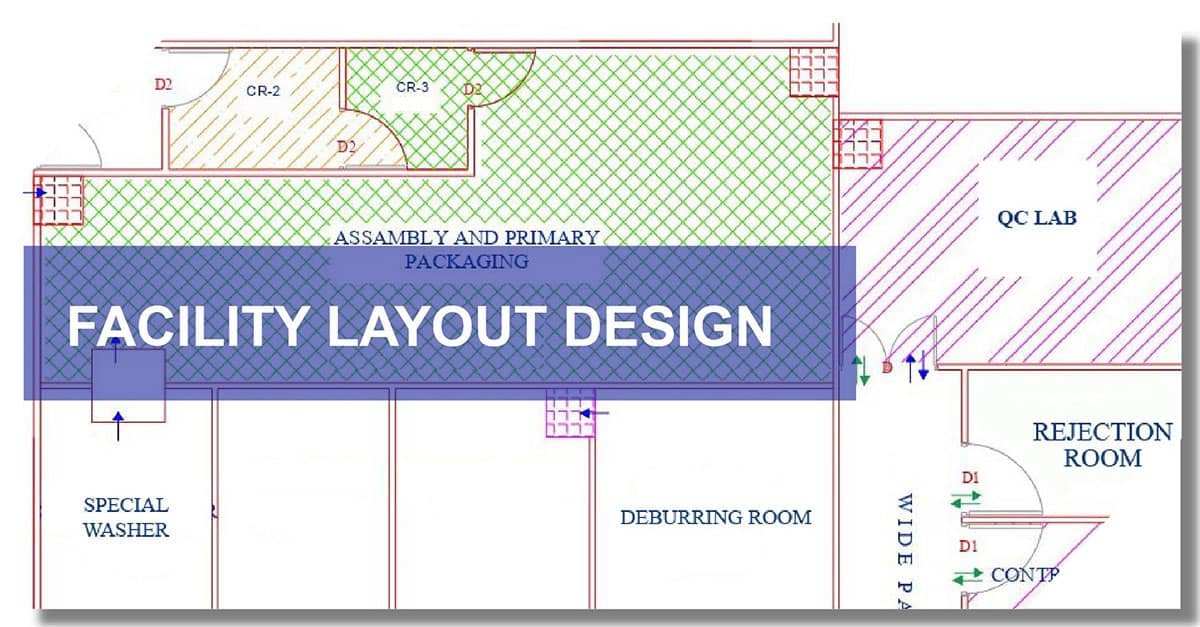
Our expert team at Operon Strategist guides manufacturers through the complexities of facility design, ensuring that every square meter of your unit is optimized. Our approach focuses on several critical pillars:
- Unidirectional Man & Material Flow: We design layouts that ensure a logical, one-way flow of personnel and materials. This is essential to prevent cross-contamination and eliminate operational bottlenecks.
- Strategic Product Segregation: By defining dedicated zones for different processes, we maintain high quality-control standards and prevent product mix-ups.
- Cleanroom & Classified Area Integration: We specialize in designing cleanrooms and classified areas that meet the rigorous environmental standards required for advanced medical device production.
- Precision AutoCAD Planning: Our AutoCAD experts translate your architectural inputs into a compliant technical design, determining exact space requirements for machinery and equipment.
Scalability & Future-Proofing: We consult on expansion plans from day one, ensuring your facility can grow or adapt to new product lines without requiring a total redesign.
Why is Manufacturing Plant Layout Design Critical for Medical Devices?
In the medical device sector, a facility’s layout is much more than a simple floor plan; it is the physical manifestation of your quality management strategy. Because precision and regulatory compliance are the cornerstones of this industry, a professionally engineered plant layout serves as the essential starting point for any successful operation.
Beyond simply organizing machinery, an optimized design integrates efficiency with safety by establishing a logical sequence of operations. This strategic approach ensures that every step of the manufacturing process aligns with stringent global standards, such as those set by the US FDA and cGMP, effectively building quality into the infrastructure itself. By focusing on the seamless flow of both personnel and materials, manufacturers can drastically reduce the risk of cross-contamination and the operational errors that often lead to costly regulatory delays.
Ultimately, investing in a robust facility design simplifies the path to market by ensuring constant audit readiness and minimizing compliance risks. It provides the structural integrity needed to scale production and adapt to new technologies, positioning a business for sustained growth and a decisive competitive edge in an increasingly demanding global marketplace.
Advantages of Professional Manufacturing Plant Layout Design Services for Medical Devices
- Seamless Compliance: Effortlessly meet global standards, including US FDA, CE Marking, and GMP regulations.
- Optimized Efficiency: Streamline operations to minimize downtime and maximize output.
- Boosted Productivity: Achieve faster production cycles while maintaining top-notch quality.
- Exceptional Quality Control: Implement robust systems to ensure consistent product excellence.
- Uncompromised Safety: Design layouts that prioritize the well-being of staff and product integrity.

Step By Step Process for Manufacturing Plant Layout Design for Medical Devices:
Designing the perfect manufacturing plant layout for medical devices involves careful planning, ensuring regulatory compliance, cleanliness, and efficient workflows. Here’s an easy-to-follow process to guide you:
- Understand Your Manufacturing Process: Get familiar with every stage of production.
- Map Out Your Workflow: Plan a smooth, efficient process from start to finish.
- Ensure Regulatory Compliance: Adhere to all necessary standards, like US FDA, CE Marking, and GMP.
- Create a Strategic Layout: Design your plant layout for optimal performance.
- Maximize Space Usage: Use available space efficiently to boost productivity.
- Prioritize Safety: Implement safety measures for your team and products.
- Collaborate with Experts: Work with engineers and architects for precision.
- Evaluate and Improve: Regularly assess and improve the layout to stay compliant and efficient.
Following this process will help you build a compliant and efficient facility designed for medical device manufacturing success.

As experienced medical device regulatory consultants, our manufacturing plant layout design is appreciated by various regulators for their compliance and for minimal man and material movements and product segregation. We also provide medical device consultation for India, Saudi Arabia, the USA, the UK, South Africa, Oman, Iran & Egypt.
Collaborate with a team that brings comprehensive industry knowledge and a proven track record of driving manufacturing success.
Ready to build your facility? Get in touch with us today to kickstart your medical device project with our expert insights!
By combining our global technical expertise with a deep understanding of local market dynamics, Operon Strategist serves as a premier partner for medical device manufacturers looking to establish world-class facilities. Here is why industry leaders choose us for their manufacturing setup and layout design needs:
Why Choose Operon Strategist for Your Medical Device Manufacturing Success?
Choosing Operon Strategist means opting for a partnership rooted in quality, precision, and operational excellence. We provide end-to-end support for manufacturers—with specialized expertise in the Costa Rican medical hub—ensuring every facility we design is audit-ready and optimized for peak performance.
- Specialized Technical Expertise: We specialize in creating high-performance, optimized layouts specifically engineered for the complex demands of medical device production.
- Global Regulatory Compliance: Our designs ensure your facility meets all international mandates, including FDA and ISO standards, while simultaneously boosting efficiency.
- Bespoke & Strategic Design: We don’t believe in one-size-fits-all. Our team crafts personalized layout solutions based on a comprehensive evaluation of your unique product requirements and site challenges.
- Seamless End-to-End Implementation: From the initial architectural concept to the final equipment installation, we provide hands-on support to ensure a smooth transition to full-scale manufacturing.
- Enhanced Operational Performance: Our layouts are designed to streamline workflows, guarantee personnel safety, and drive overall plant productivity for long-term success.
- Data-Driven Review Process: Before we design, we conduct a thorough audit of your current or planned infrastructure to pinpoint key areas for technical and financial enhancement.
FAQ
What is required to set up a medical device manufacturing facility in Costa Rica?
Setting up a medical device manufacturing facility requires compliance with regulatory standards, facility layout design, equipment installation, cleanroom setup, validation, and certification processes.
What regulatory standards are followed for medical device facilities?
We ensure compliance with international medical device regulations, including FDA 21 CFR Part 820, ISO 13485, CE Marking, and Costa Rica’s health authority requirements.
How long does it take to set up a medical device manufacturing facility?
The setup timeline varies depending on the project scope, but our streamlined approach ensures regulatory-compliant execution within an optimized timeframe, typically ranging from a few months to a year.