Clean Room Design Consultant for Medical Devices
Regulatory Aspects of Clean Room Design
As a Clean Room Design consultant, we provide expertise clean room design solution to medical device manufacturers. Clean Room is the facility ordinarily utilized as a part of specialized industrial production or scientific research.

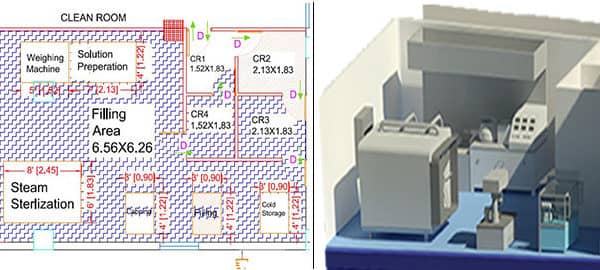
What is a Medical Device Clean Room?
A clean room is a specialized, enclosed environment within a manufacturing facility designed to strictly control air pollutants, humidity, and personnel access. By minimizing environmental contaminants—such as dust, airborne microbes, aerosol particles, and chemical vapors—clean rooms ensure the sterility and safety of medical devices.
As an expert Clean Room Design Consultant, Operon Strategist provides comprehensive design, validation, and regulatory solutions tailored to international standards. We ensure your facility operates efficiently and complies with ISO 14644-1, delivering the precise environmental control necessary for high-quality, safe medical device production.
Why Medical Device Manufacturers Need Clean Rooms
Clean rooms are more than just sterilized spaces; they are high-performance environments engineered to eliminate dust, microbes, and aerosol particles. For medical device manufacturers, these controlled environments are critical for ensuring product reliability and patient safety.
- Minimized Contamination: Maintaining ultra-low pollutant levels prevents micro-defects and ensures the structural and functional integrity of sensitive devices.
- Regulatory Compliance: Clean rooms are essential for meeting stringent global mandates, such as ISO 14644-1 and other international sterility standards.
- Enhanced Product Safety: By controlling the manufacturing atmosphere, you guarantee the sterility, efficacy, and consistent quality of every device produced.
- Strategic Project Planning: Professional clean room design allows manufacturers to visualize the full project scope and total costs before construction begins, preventing expensive mid-project adjustments.
- Operational Efficiency: When correctly designed and built, clean rooms act as “execution machines,” streamlining workflows while providing a total facility solution that requires minimal client maintenance.
Looking for Consultant?
Clean Room Design Requirements for Medical Devices
To ensure optimal performance, clean rooms must meet specific requirements:
- Air Quality: Use HEPA filters to remove particles as small as 0.3 microns; maintain positive air pressure to prevent contamination.
- Temperature & Humidity Control: Maintain 18–26°C and relative humidity between 30–70%.
- Gowning Procedures: Mandate cleanroom garments and controlled entry/exit to reduce contamination from personnel.
- Layout Design: Include designated zones for gowning, storage, sterilization, and waste disposal to prevent cross-contamination.
- Surface Cleanliness: Use smooth, crack-free surfaces for easy cleaning and disinfection.
- Sterilization Areas: Dedicated zones for sterilizing medical devices to maintain product safety.
- Monitoring Systems: Regularly test air quality, temperature, and microbial levels to ensure ongoing compliance.
Regulatory Guidance for Clean Rooms
Operon Strategist ensures your clean room design meets regulatory standards across countries such as Costa Rica, South Africa, Egypt, Oman, UK, USA, Saudi Arabia, and Iran. We provide guidance on:
- Entry and exit protocols
- Gowning and de-gowning procedures
- Air circulation systems to maintain ISO compliance
- Regulatory approvals and documentation support
How Operon Strategist Supports Your Clean Room Design and Validation
As a premier Clean Room Design Consultant, Operon Strategist provides end-to-end expertise for setting up new manufacturing units or upgrading existing facilities. We deliver tailored design and validation services that strictly adhere to international norms and regulatory standards.
Our Clean Room Consulting Expertise
- Custom Design & AutoCAD Services: We provide precision AutoCAD layouts tailored to manufacturing, scientific labs, R&D, and primary packaging. Our designs optimize airflow and contamination control for maximum efficiency.
- Comprehensive Validation & Compliance: We guide you through the full ISO 14644-1 lifecycle, managing everything from installation to operational qualification (IQ/OQ/PQ) to ensure a sterile production environment.
- Regulatory & HVAC Integration: Our team ensures your facility meets stringent FDA, EU MDR, and local standards. We specialize in integrating specialized HVAC systems designed for sterile medical device production.
- Global Project Success: With a proven track record of successful clean room completions, we offer the technical and regulatory support necessary to transform your facility into a high-performance “execution machine.”
Operon Strategist’s Role in Clean Room Design and Regulatory Approval
As specialized Clean Room Design Consultants, Operon Strategist serves as a strategic partner for medical device manufacturers, guiding them through the complex journey from initial conceptualization to full regulatory approval. Our role is to ensure that your facility is not only physically optimized for production but also fully compliant with international air quality standards.
Strategic Design and Technical Oversight
- Conceptualization and Planning: We manage the design and layout phases, ensuring the structure supports ideal air quality levels. This includes engineering the entire air circulation framework and downstream air returns—such as low-wall returns in vertical flow rooms—to maintain particle counts according to ISO 14644-1.
- Document and Plan Review: Before construction begins, we conduct rigorous surveys of plans and specifications to identify and correct design errors, saving time and preventing costly rework.
- Operational Procedures: Beyond the physical walls, we define the “supporting elements” of a clean room, including strict entry-exit protocols and gowning procedures essential for maintaining environmental integrity.
Commissioning and Environmental Control
- Building Commissioning: We provide commissioning services to guarantee that the finished facility operates exactly as intended in the initial design phase.
- Environmental Parameter Management: Our experts optimize the control of humidity, temperature, and pressure, providing recommendations for short-term goals and long-term facility upgrades.
Global Regulatory and QMS Guidance
Operon Strategist is a leading regulatory consultancy that works closely with authorities such as SFDA. We ensure your clean room design integrates seamlessly into a broader Quality Management System (QMS), supporting your strategic development.
We have a proven track record of guiding manufacturers and service providers in Costa Rica, and we extend our specialized consultation services to India, Saudi Arabia, the USA, the UK, South Africa, Oman, Iran, and Egypt.
FAQ's
How are clean rooms classified as per ISO Standards?
Medical device manufacturing typically performs in an ISO 5 (Class 100) to ISO 8 (Class 100,000) cleanroom. Medical device packaging typically is conducted in an ISO 7 (Class 10,000) or ISO 8 (Class 100,000) cleanroom with an ISO 8 (Class 100,000) gowning room.
What standards apply to clean rooms for medical devices?
Clean rooms must comply with ISO 14644, ISO 13485, Good Manufacturing Practices (GMP), and industry-specific regulatory guidelines.
What are ISO standards for clean rooms?
The ISO 1 specification for cleanrooms requires less than 2 particles greater than 0.3 microns and no particles greater than 1.0 microns per cubic meter. The ISO 2 specification for cleanrooms requires less than 11 particles greater than 0.3 microns and no particles greater than 1.0 microns per cubic meter.
What are the different ISO 14644 clean room classifications?
ISO 14644 defines clean room classifications from ISO Class 1 (highest cleanliness) to ISO Class 9 (lowest cleanliness), depending on airborne particle concentration limits.