Medical Device Validation Documentation
What is Medical Device Process Validation?
Medical Device Process Validation is the systematic collection and evaluation of data—from initial design through full-scale production—to provide scientific evidence that a manufacturing process will consistently deliver high-quality products. In essence, it establishes documented proof that your procedures reliably maintain the required levels of safety and compliance at every stage.
Rather than relying solely on final product testing, validation proves that the manufacturing process itself is stable and capable of meeting pre-defined specifications every time. This rigorous documentation is a mandatory requirement for global regulatory alignment, including FDA 21 CFR Part 820, ISO 13485:2016, and EU MDR. By implementing robust validation, manufacturers mitigate the risk of recalls, enhance operational efficiency, and provide clinicians with absolute confidence in device efficacy.
Let's Grow Your Business Together
The Critical Role of Utilities, Equipment, and Process Validation
In the medical device lifecycle, Verification and Validation (V&V) serve as the primary safeguards for both patient safety and manufacturer integrity. Verification ensures that the device design strictly adheres to all engineering specifications, while validation confirms that the device and its manufacturing processes perform exactly as intended in real-world clinical scenarios.
Beyond a simple regulatory hurdle, a rigorous validation plan for utilities and equipment is essential for several strategic reasons:
- Guaranteed Safety and Efficacy: It ensures that every device produced is safe for the end-user and functions as designed.
- Global Market Access: Comprehensive V&V documentation is a prerequisite for maintaining compliance with international regulatory standards, facilitating smoother market entry.
- Documented Consistency: It provides objective, scientific proof that production processes yield consistent quality, batch after batch.
- Risk and Cost Mitigation: By identifying potential flaws early, manufacturers can minimize production risks and avoid the catastrophic financial and reputational damage of product recalls.
- Operational Efficiency: Validated equipment and utilities lead to more predictable production cycles, reducing downtime and optimizing resource utilization.
Our team specializes in executing detailed validation testing and V&V planning, ensuring your manufacturing setup meets the highest global benchmarks for quality and reliability.
What Is the Difference Between Process Verification and Validation?
Medical Device Process Validation is the framework that ensures every stage of your production consistently yields safe, effective, and high-quality devices. By combining the inspection of individual products with the validation of entire systems, manufacturers can satisfy stringent regulatory mandates like FDA 21 CFR Part 820 and ISO 13485, while simultaneously reducing operational risks.
- Process Verification: This involves the inspection or testing of individual units to ensure they align with specific design requirements. It is best suited for low-volume or straightforward devices where testing every unit is feasible to prove compliance with safety standards.
- Process Validation: This confirms that the entire manufacturing process is capable of consistently producing compliant and safe devices. This is essential for complex or high-volume production where testing every single item is impractical, ensuring long-term process reliability and regulatory approval.
Key Difference: Verification focuses on products, while validation focuses on the process.
What Are the Different Types of Process Validation?
Process validation plays a crucial role at different stages of the production lifecycle, and understanding the various types helps ensure your products meet quality standards every step of the way. Here are the key types of process validation:
- Prospective Validation
- Concurrent Validation
- Retrospective Validation
- Revalidation
Each type serves a unique purpose in ensuring your processes remain reliable, efficient, and compliant with regulatory standards.

Medical Device Process Validation Services
To maintain consistent quality and meet regulatory standards, Medical Device Process Validation is essential for every manufacturer. It ensures that your processes are compliant and reliable at every stage of production. Our expert validation services include:

Our Expert Validation Services is Divided into the Following Sub-sections:
- HVAC Validation
- Equipment Validation
- Process Validation
- Facilities Validation
- Cleaning Validation
- Analytical Method Validation
- Personnel Validation
- Packaging Validation
- Computer System Validation
We focus on creating clear, actionable protocols and criteria for each validation step. From the initial qualification to routine monitoring, we provide detailed reports to ensure your processes run smoothly, efficiently, and in full compliance, all while maintaining product quality at every stage.
Why Do Medical Device Manufacturers Need a Validation Process?
In the medical device industry, validation is not just a regulatory hurdle; it is a critical strategy for ensuring product integrity and patient safety. Implementing a robust validation process allows manufacturers to:
- Meet Global Regulatory Standards: Validation is a mandatory requirement for complying with international regulations such as FDA 21 CFR Part 820 and ISO 13485:2016. It ensures every product adheres to strict safety and quality guidelines necessary for legal market access.
- Provide Objective Proof of Quality: It generates the documented evidence required to prove that a device consistently meets its design specifications and user needs. This is achieved through structured testing and rigorous inspection protocols.
- Ensure Reliable Real-World Performance: Validation confirms that a device will perform as intended under actual clinical conditions. This guarantees that the device remains safe, effective, and functional when it reaches the end-user.
- Achieve and Maintain FDA Compliance: A thoroughly validated process is essential for FDA approval. It demonstrates to regulatory bodies that both the product and the manufacturing methods strictly follow established Quality System Regulations (QSR).
- Strengthen Process Control and Consistency: Before a device is launched, validation ensures that production processes are stable and repeatable. By establishing this control, manufacturers minimize the risk of defects, reduce the likelihood of costly recalls, and ensure consistent outcomes across every batch.
How Operon Strategist Supports Your Medical Device Process Validation
Operon Strategist offers end-to-end medical device process validation support, ensuring that all activities—from initial planning to final execution—are conducted with precision and rigorous documentation. By partnering with us, manufacturers gain access to a team of experts dedicated to building robust, audit-ready systems that stand up to global scrutiny.
Our Comprehensive Validation Services
- Tailored Validation Strategies: We develop customized protocols that match your specific product type and manufacturing process, ensuring full alignment with international quality standards.
- Simplified Regulatory Compliance: Our consultants navigate the complexities of FDA 21 CFR Part 820, ISO 13485:2016, and EU MDR, providing you with a clear and confident path to compliance.
- FDA-Compliant Protocols & Reports: We prepare structured, audit-ready documentation, including Design Qualification (DQ), Installation Qualification (IQ), Operational Qualification (OQ), and Performance Qualification (PQ).
- Lifecycle Expertise: From equipment and utility qualification (HVAC, water systems, etc.) to specialized packaging and manufacturing process validation, we ensure every link in your production chain is secure.
- Holistic Consulting Support: Whether you are validating a new facility or upgrading existing processes, we deliver the expertise needed to ensure total traceability and operational efficiency.
Why Choose Operon Strategist as Your Validation Partner?
Choosing Operon Strategist means leveraging a global wealth of experience. Our experts guide you through every critical milestone, including site acceptance testing, facility qualification, and design validation.
Our global presence gives us a unique advantage in working with diverse regulatory bodies. In the Costa Rica region, our growing base of satisfied customers is a testament to our commitment to quality and local market expertise. Furthermore, we provide specialized medical device consultation across India, Saudi Arabia, the USA, the UK, South Africa, Oman, Iran, and Egypt.
Partner with us to ensure your manufacturing processes are market-ready and fully compliant. Contact us today to discuss your validation requirements.
Key Elements of a Validation Plan for Medical Devices
A well-structured validation plan is essential for the medical device validation process. It includes guidelines for conducting validation activities, identifying protocols, and detailing the required tests. At Operon Strategist, we offer expert guidance on creating a comprehensive plan tailored to your specific product and market needs.
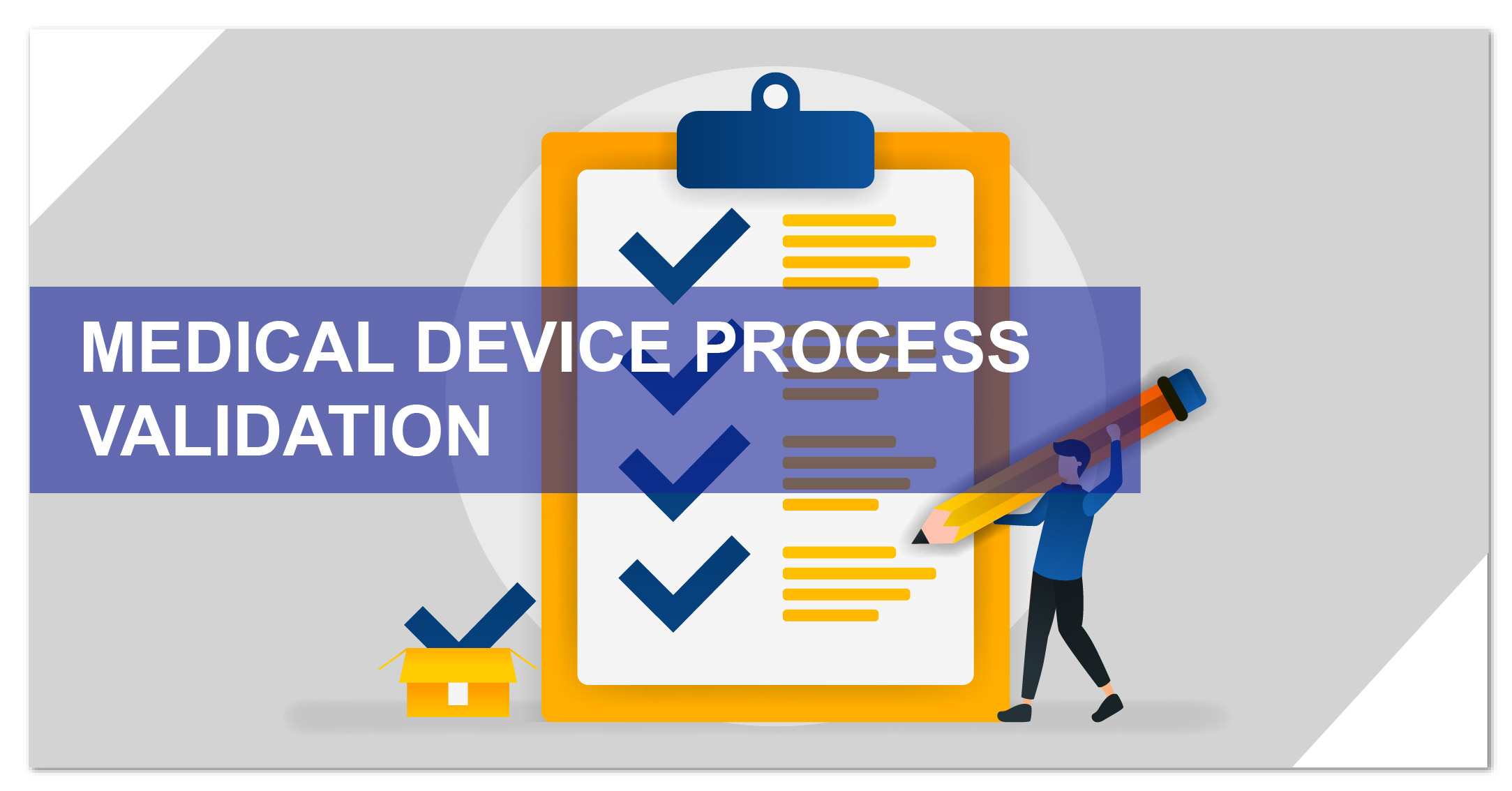
What are the 3 Stages of Process Validation?
Process validation consists of three legally enforceable stages:
- Stage 1 – Process Design: Defining the manufacturing process based on development and scale-up data.
- Stage 2 – Process Qualification: Evaluating the facility, utilities, and equipment to ensure the design is capable of reproducible commercial manufacturing.
- Stage 3 – Continued Process Verification: Providing ongoing assurance during routine production to ensure the process remains in a state of control.

What is a Process Validation Protocol in Medical Devices?
A Process Validation Protocol is a formal, documented plan that serves as a blueprint for how validation activities will be executed. It defines the specific objectives, testing parameters, equipment to be used, and the predetermined acceptance criteria for a manufacturing process. This document is essential for demonstrating that a process is stable and capable of consistently producing medical devices that meet their intended quality and safety requirements.
How long does it take to complete validation documentation?
The timeline typically ranges from a few weeks to several months. This duration is influenced by the complexity of the medical device, the scale of the manufacturing process, and specific regulatory requirements (such as FDA or EU MDR). Having a stable production environment and clear design specifications can significantly accelerate the process.
What is validation documentation in medical device manufacturing?
Validation documentation is the formal evidence—including protocols, test reports, and compliance records—proving that equipment, software, and processes consistently meet quality standards. It serves as a permanent, audit-ready record that your manufacturing system is stable and reliable.
Why is validation documentation critical for regulatory compliance?
Regulatory bodies like the FDA and ISO require this documentation to verify that a device meets safety and efficacy standards before it reaches the market. It is the essential proof that your product is manufactured under controlled, repeatable conditions, which is mandatory for legal approval.
What is Medical Device Process Qualification?
Process Qualification (PQ) is the stage where the manufacturing environment, utilities, and equipment are verified to function effectively and reproducibly. It is critical for identifying and mitigating production risks, ensuring that every device consistently meets ISO 13485 and FDA quality standards throughout its lifecycle.