Manufacturing Facility Setup for Medical Device Companies in the UK
Setting up a medical device manufacturing facility in the UK involves a lot more than just space and equipment. You need a well-planned layout, regulatory approvals, process validation, and cleanroom compliance to meet MHRA and ISO 13485 requirements. That’s where we come in.
As experienced medical device turnkey project consultants, we help UK-based startups and manufacturers build fully compliant, functional, and audit-ready manufacturing facilities from the ground up.
What Is a Manufacturing Facility for Medical Devices?
A medical device manufacturing facility is a controlled environment where medical devices are produced, assembled, and packaged in compliance with regulatory and quality standards. It includes dedicated zones for production, quality control, cleanroom operations, and storage, all designed to ensure product safety and consistency.
Why Is a Manufacturing Facility Important for Medical Devices?
The quality and safety of medical devices depend heavily on the facility in which they are made. A well-designed manufacturing setup:
- Ensures compliance with MHRA and international regulations
- Supports consistent product quality and traceability
- Minimizes contamination risks through proper material flow and cleanroom standards
- Enhances productivity through efficient layout and automation
Looking for Manufacturing Plant Layout Design Consultant?
Fill the Form or Mail Us to: enquiry@operonstrategist.com
Why You Need a Turnkey Project Consultant?
Setting up a compliant facility requires deep knowledge of the UK medical device regulatory landscape, technical expertise, and project coordination. We simplify the process and reduce delays by handling all aspects for you—planning, design, execution, and documentation.
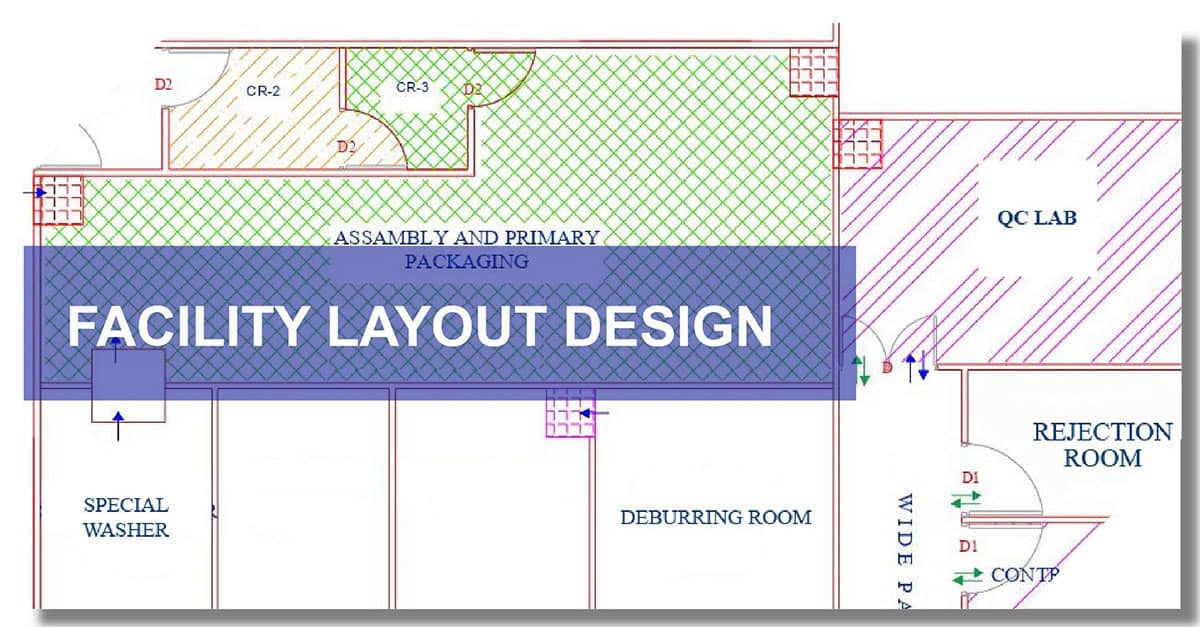
Process of Manufacturing Facility Design
Our approach to designing a manufacturing facility for medical devices involves the following steps:
- Requirement Assessment – Understanding your device type, classification, and production volume.
- Site Evaluation – Selecting a location and assessing feasibility based on space, access, and utilities.
- Layout Planning – Designing optimal workflows for material movement, personnel, and production processes.
- Cleanroom & HVAC Design – Planning controlled environments that meet ISO 14644 standards.
- Utility Planning – Ensuring reliable power, water, air supply, and waste management.
- Compliance Planning – Integrating regulatory needs into facility design from the start.
Our End-to-End Manufacturing Facility Setup Services
We manage the full setup of your medical device manufacturing facility, tailored to your product and regulatory needs:
- Site selection guidance and feasibility study
- Layout planning and material flow design
- HVAC and cleanroom design (ISO 14644 compliance)
- Equipment selection and validation
- Quality Management System (QMS) setup aligned with ISO 13485
- Documentation support for UKCA, CE marking, and MHRA approvals
- Staff training and handover

Compliance We Help You Achieve
Ready to set up your medical device manufacturing unit in the UK? Let Operon Strategist’s turnkey consultants handle the complexity, so you can focus on innovation and growth.
Contact us today for a free consultation or to discuss your project needs.
What Makes Us Different
- Years of experience with UK and global medical device regulations
- Customised approach based on your device class and business model
- Full documentation and regulatory support
- On-site assistance during audits and inspections