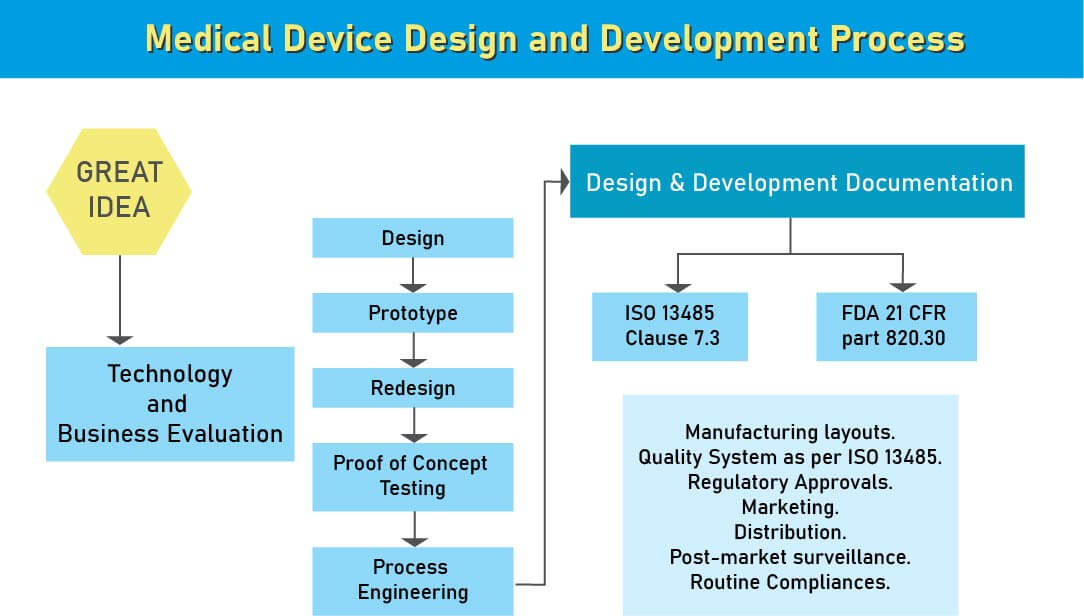
Design and Development Documentation
Understanding Medical Device Design and Development
Design & development documentation is essential for ensuring the safety, performance, and regulatory compliance of medical devices. A poorly documented design process can lead to regulatory setbacks, product recalls, and quality issues under standards like ISO 13485, EU MDR, SFDA, and USFDA. Operon Strategist supports manufacturers in creating robust, compliant design files that align with global requirements and ensure successful market entry.

Importance of Medical Device Design and Development
- Enhances Patient Care
Proper design improves clinical outcomes by ensuring safety, effectiveness, and usability in real-world healthcare settings. - Addresses Unmet Medical Needs
Design innovation helps solve specific clinical challenges and supports the development of specialized devices. - Improves Usability and Reduces Errors
User-centered design simplifies operation, reduces human error, and improves healthcare worker efficiency. - Ensures Regulatory Compliance
Aligns with international standards like ISO 13485:2016 Clause 7.3 and FDA 21 CFR Part 820.30, supporting global market access. - Drives Innovation and Technological Advancement
A strong design foundation enables next-generation solutions, such as smart devices, wearable technologies, and minimally invasive tools. - Supports Global Market Success
Well-designed products are more likely to meet both regulatory approval and user expectations, improving adoption and market reach.
The Medical Device Design And Development Process Guide
During the design and development stage of the medical device design process, we assist various medical device manufacturing industries in UK to ensure that appropriate steps are taken to meet the regulatory compliance of the medical device design and development. After analyzing a new medical device, the next step in its product development is the medical device design. This is the most important stage in medical device development for a flawed design may ahead of it being ineffective or risky. At the medical device design stage, a design control process system is required.
Being, design controls are simple and logical steps to ensure that what you develop is what you determine to develop and that the final product meets your customer’s needs and expectations.
- Medical device development uses a ton of comparative parts in a large number of various medical devices. A solid definition extricated by dissecting the market needs. When you’re finished with the products definition and thought, you have to consider systems like FDA has characterized and licensed innovation rights. Medical devices classification depends on the hazard related to the utilization and upheld by law. So as to get into the market, the medical devices need to go through certain administrative compliances, subject to both provincial and worldwide guidelines. Medical devices measures are useful and upheld by law in indicating and assessing the prerequisite for structure and execution parameters for biomedical materials, apparatuses, and gear. These medical devices standards permit establishments in the medical device field, for example, products manufacturers, research centres, and others to review and survey such hardware and devices to guarantee standard quality and ease of use.
- The International Organization for Standardization likewise have details for medical devices principles. ISO 13485 is broadly utilized guidelines over the world for medical devices quality administration. Other than these worldwide models, there are sure gauges which are area explicit and every one of them are embraced from universal norms with little adjustment and constraint.
- Medical devices manufacturers need to pursue Design Control rules since the administrative bodies like FDA, European Commission, and others need to guarantee that the medical devices are alright for potential clients before makers begin to advertise the devices. Beginning stage from which Design Control starts is Design Input advancement and endorsement, which comprises of device design and manufacturing procedures to be completed in the generation stage. Design control is a comprehensive methodology and doesn’t end with moving the design to the generation stage when the plan is settled. It additionally affects manufacturing procedures as indicated by the adjustments in the design stage or even after creation input. It is a progressing procedure to build up a product that is usable for a client and in this way for the improved product, it considers progressive changes from utilization design just as breaking down failed items.
What is Product Development in Medical Devices?
Medical device product development is the complete process of bringing a medical device from concept to market. It begins with identifying a clinical need or market opportunity and continues through market research, concept design, engineering, regulatory compliance, manufacturing, and commercialization.
This process also includes post-market surveillance to ensure ongoing safety and performance. Each stage is crucial to creating a compliant, effective, and user-friendly product that meets both regulatory standards and healthcare demands.
What Do You Mean by Design Control in Medical Devices?
Design control refers to a structured and regulated process required by the FDA (21 CFR Part 820.30) for the development of Class II and Class III medical devices. It ensures that every stage of the device’s design meets safety, performance, and regulatory standards through documented evidence.
This process involves detailed planning, risk management, design inputs and outputs, verification, validation, and design reviews. For medical device companies, proper device classification is essential to determine if design controls apply, making compliance a critical part of product development.
How to Design and Develop a Medical Device?
It takes a significant amount of effort to deliver the right healthcare solution that meets customer demands. A right healthcare solution demands everybody staying on the same page, with strong scope definition from end user’s need, collaborative efforts across the team, adherence to specification and requirements extracted from product definition, simultaneously mitigating risks and sticking to the best possible quality.
Medical Device Design Services & Product Development Process?
- Feasibility
- Planning
- Design and development
- Verification
- Validation
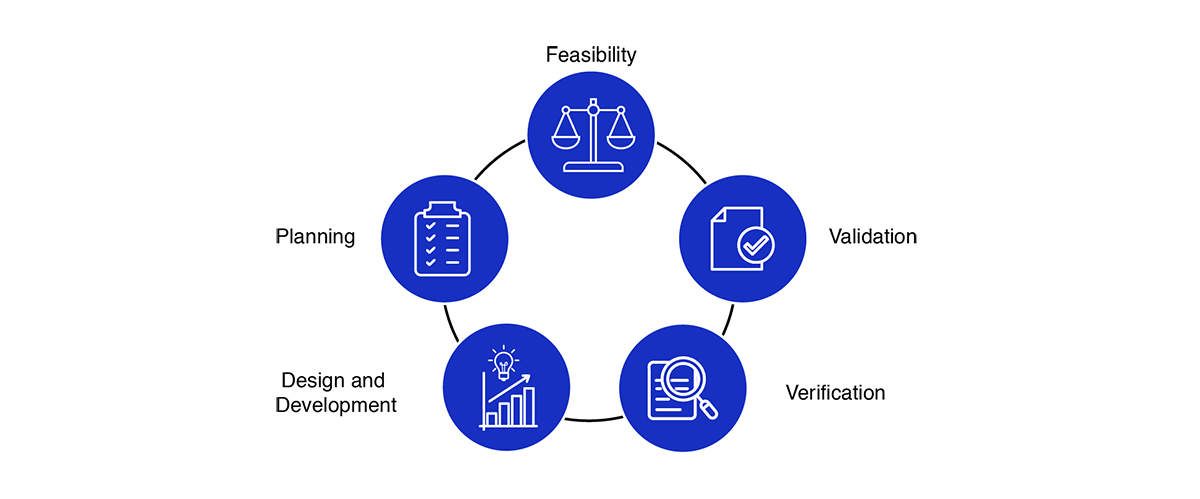
Medical device design services and development process encourages an early focus on clear problem definition and de-risking a wide variety of potential solutions. By later phases, the funnel of medical device design options narrows significantly, converging on a final product that has been thoroughly shown to meet the customer needs and is ready for distribution.
The Medical Device Design and Development Services Includes :
Combination Product - Drug - Device
Each manufacturer of Drug Device combination product (e.g. Drug, device combination products like prefilled syringes, applicators of the tropical products) shall have adequate design and development activity done so as to prove the adequacy of the safety and efficacy of the product. The medical device design and development activity is the systematic methodology, which establishes the proper medical device design and development.
Medical Device Design Control
After conceptualizing a new medical device, the next step in its product advancement is the design. This is the most important stage in the advancement of a medical device since a defective plan may prompt it being inadequate or dangerous (that is, not affirmed or cleared by the administrative organization). At the medical device design stage, an outline control process should be started and actualized as a feature of the quality system requirement.
Our Role in Device Design Control
At Operon Strategist, we support medical device manufacturers in achieving regulatory compliance through effective design control implementation. As experienced medical device design consultants, we guide your team through the full process validating tools, training personnel, and helping establish a qualitative Quality System Management (QSM) aligned with ISO 13485 and FDA 21 CFR 820.30.
With strong industry partnerships and a deep understanding of global regulations, we offer cost-effective, error-free, and timely solutions tailored to your development goals. Whether you’re in the early design phase or preparing for market approval in the UK or internationally, you can rely on us to ensure a compliant and efficient design control process.
Get Expert Consulting for Medical Device Design & Development
FAQs
What is medical device design and development?
It’s the process of transforming a concept into a safe, effective, and compliant medical device through structured planning, testing, and validation.
Why is design control important?
Design control ensures regulatory compliance (ISO 13485, FDA 21 CFR 820.30), product safety, and reduced risk of failures or recalls.
Who must follow design control regulations?
Manufacturers of Class II and III medical devices, and some Class I devices depending on market regulations (EU, UK, US, etc.).
How does Operon Strategist support this process?
We offer expert consulting for design planning, documentation, validation, and compliance with global medical device regulations.
What causes most product failures?
Poor design planning, weak risk control, and lack of validation are key reasons for product recalls and non-compliance.