Medical Device Manufacturing Facility Setup in the Netherlands
Establishing a compliant, efficient, and future-ready medical device manufacturing facility is critical to your product’s success in the European and global markets. Operon Strategist provides end-to-end support for setting up turnkey medical device manufacturing facilities in the Netherlands—designed to meet EU MDR, ISO 13485, CE marking, and local Dutch regulatory requirements.
What is Medical Device Manufacturing Facility Layout?
A medical device manufacturing facility layout refers to the strategic planning and design of physical spaces, equipment placement, workflows, and clean zones within a production unit to ensure efficient, safe, and regulatory-compliant manufacturing of medical devices.
This layout is not just about organizing space—it’s about optimizing production flow, minimizing contamination risks, and meeting stringent international standards like ISO 13485, ISO 14644 (cleanroom classification), and EU MDR or FDA 21 CFR Part 820 requirements.
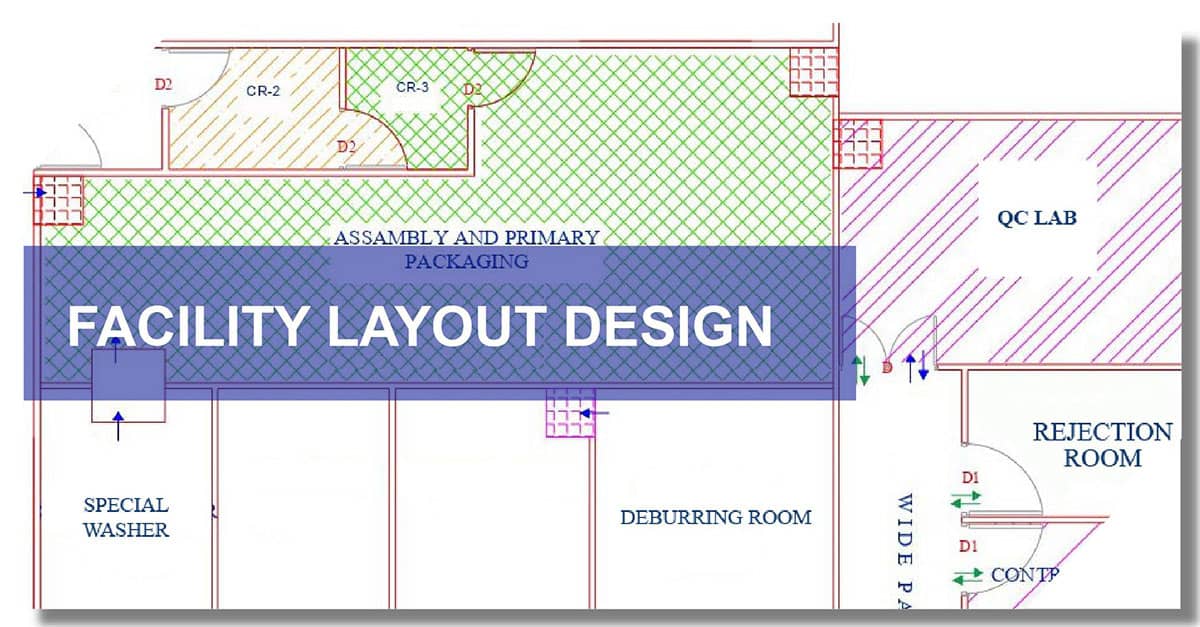
Key Components of a Medical Device Facility Layout
- Raw Material Storage Area: Controlled environment for storing incoming materials with segregation for quarantine, approved, and rejected items.
- Cleanroom Zones (ISO Class 7/8 or higher): For aseptic or sterile product manufacturing, with controlled airflow, temperature, humidity, and particulate matter.
- Manufacturing and Assembly Area: Designed for smooth unidirectional flow of materials and personnel to prevent cross-contamination.
- In-Process QC and Final Testing Areas: Designated zones for quality inspection, functional testing, and documentation.
- Sterilization Area (if applicable): For processes like ETO, gamma, or steam sterilization with proper exhaust and validation protocols.
- Packaging and Labeling Zone: Separate from manufacturing to ensure labeling accuracy and prevent mix-ups.
- Finished Goods Storage: Temperature and humidity-controlled area for storing products ready for shipment.
- Personnel Movement Areas: Gowning/de-gowning zones, airlocks, and pass-through chambers to maintain cleanliness and avoid contamination.
- Utilities and Equipment Rooms: For housing HVAC, compressed air, purified water systems, etc., without disrupting core operations.
Looking for Manufacturing Plant Layout Design Consultant?
Fill the Form or Mail Us to: enquiry@operonstrategist.com

Why is Manufacturing Plant Layout Design Important for Medical Devices?
Designing a manufacturing plant layout for medical devices is more than just organizing spaces—it’s about meeting strict regulations and ensuring quality at every step. For medical device manufacturers in the Netherlands, a well-planned layout boosts productivity, improves operational flow, and guarantees compliance with European standards. By prioritizing efficiency and adhering to regulatory requirements, your facility becomes a foundation for producing safe, high-quality medical devices. Partner with professionals to create a layout that works for you and meets industry demands.
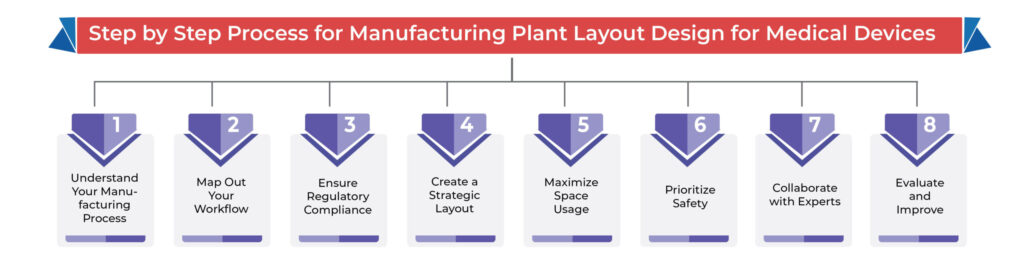
Step By Step Process for Manufacturing Plant Layout Design for Medical Devices:
Importance of an Optimized Layout
- Ensures compliance with EU MDR, FDA, ISO 13485, and GMP
- Improves production efficiency and reduces downtime
- Facilitates cleanroom validation and regulatory audits
- Enhances product quality and patient safety
- Supports scalability and future expansion
Why Choose Operon Strategist?
Operon Strategist is your trusted partner for setting up regulatory-compliant, efficient medical device manufacturing facilities in the Netherlands and across the EU.
- Regulatory Expertise: We align your facility with ISO 13485, ISO 14644, EU MDR, and CE marking—ensuring you’re audit-ready.
- Turnkey Solutions: From layout design and cleanroom setup to QMS implementation and validation—we handle it all.
- Tailored Facility Design: Custom layouts based on your device type, risk class, and production needs—for optimal compliance and efficiency.
- Cross-Functional Team: Our engineers, regulatory consultants, and QMS experts work together to deliver seamless project execution.
- Proven Track Record: 100+ successful projects worldwide with deep knowledge of the Dutch and EU regulatory landscape.
- Documentation & Training: We support you with full validation (IQ/OQ/PQ), technical documentation, SOPs, and staff training.
Get Expert Support from Operon Strategist for Your Manufacturing Facility Setup!
FAQs
What regulatory approvals are required for a medical device manufacturing facility?
Regulatory approvals depend on the region. In the EU, compliance with MDR and ISO 13485 is required. In the US, manufacturers must follow FDA 21 CFR Part 820. Other regulations such as CE Marking, UKCA, and CDSCO (India) may also be applicable.
How do you ensure compliance with ISO 13485 in a manufacturing facility?
Compliance is achieved through proper documentation, quality control, process validation, risk management, and internal audits. Operon Strategist assists in setting up and maintaining an ISO 13485-compliant QMS.
What are the key factors in designing a medical device manufacturing plant layout?
Important factors include regulatory compliance, process flow efficiency, cleanroom integration, equipment placement, material handling, and safety measures. Operon Strategist designs layouts that optimize space utilization while ensuring compliance.
What is cleanroom validation, and why is it important?
Cleanroom validation ensures that the controlled environment meets ISO 14644 or GMP standards for particle count, airflow, temperature, and humidity. It is crucial for preventing contamination in medical device manufacturing.
How does Operon Strategist assist with medical device manufacturing facility setup?
Operon Strategist provides turnkey consulting, covering plant layout design, regulatory approvals, QMS implementation, validation, and ongoing compliance support to establish a fully compliant facility.