Introduction to MHRA Medical Device Registration
Entering the UK market requires more than just a quality product—it requires strict regulatory compliance. MHRA medical device registration is a mandatory step for any manufacturer who wants to sell medical devices or IVDs in Great Britain.
The Medicines and Healthcare Products Regulatory Agency (MHRA) is responsible for regulating medical devices in the UK. Its role is to ensure that all products placed on the market meet the required standards for safety, quality, and performance.
Without MHRA registration, a medical device cannot be legally marketed in the UK. Therefore, understanding the registration process is essential for a smooth and successful market entry.
Looking for Medical Device Regulatory Consultation?
Let’s have a word about your next project
Explore our comprehensive guide to UKCA marking for medical devices on our service page, and let Operon Strategist expertly guide you through the MHRA registration process for seamless market entry in the UK.
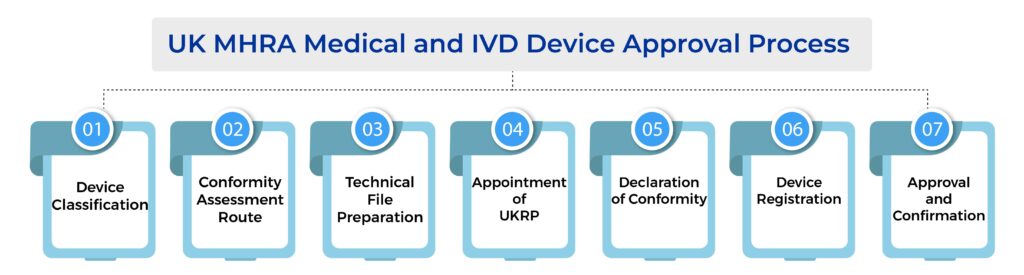
The UK MHRA Medical and IVD Device Step-by-Step Approval Process
Understanding the process step by step helps avoid delays and ensures compliance.
1. Device Classification
The first step is to determine the correct classification of your device.
- Medical Devices: Class I, IIa, IIb, III
- IVDs: Class A, B, C, D
Accurate classification is critical, as it determines the regulatory pathway and documentation requirements.
2. Conformity Assessment and QMS Implementation
Based on the classification, manufacturers must follow the appropriate conformity assessment route.
In most cases, implementing a Quality Management System (QMS) aligned with ISO 13485 is required. This ensures consistent quality and regulatory compliance.
3. Technical Documentation Preparation
A comprehensive technical file must be prepared, including:
- Device description and intended use
- Risk management documentation
- Clinical or performance evaluation
- Labeling and Instructions for Use (IFU)
Well-prepared documentation plays a key role in avoiding regulatory queries and delays.
4. Appointment of UK Responsible Person (UKRP)
Non-UK manufacturers must appoint a UKRP.
The UKRP is responsible for:
- Registering the device with MHRA
- Acting as a point of contact with regulators
- Maintaining regulatory documentation
Choosing an experienced UKRP can significantly improve the efficiency of the process.
5. Declaration of Conformity and UKCA Marking
Once compliance requirements are met, manufacturers must prepare a Declaration of Conformity and affix the UKCA marking to the device.
This step confirms that the product meets UK regulatory requirements.
6. Device Registration via DORS
The device is registered through the MHRA Device Online Registration System (DORS).
The application includes:
- Manufacturer details
- Device details
- Applicable registration fee
7. MHRA Review and Approval
After submission, MHRA reviews the application.
- Standard review time: approximately 5 working days
- Additional information may be requested if required
Once approved, MHRA issues a registration confirmation, allowing the device to be legally marketed in the UK.
Who Needs MHRA Medical Device Registration?
MHRA registration is required for:
- Medical device manufacturers
- In vitro diagnostic (IVD) manufacturers
- Non-UK manufacturers exporting to the UK
- Importers and distributors placing devices on the UK market
For manufacturers located outside the UK, appointing a UK Responsible Person (UKRP) is mandatory. The UKRP acts as a local representative and handles communication with the MHRA.
Cost of MHRA Medical Device Registration
Understanding the cost structure is important for proper planning.
Registration Fee
- MHRA charges £240 per registration
- Up to 100 devices can be included in a single submission if grouped correctly
Additional Costs
- UK Responsible Person (UKRP) services
- ISO 13485 QMS implementation
- UK Approved Body certification (if applicable)
- Technical documentation preparation
Proper planning can help control costs and avoid unnecessary expenses.
Timeline for MHRA Registration
Standard approval timeline: around 5 working days
Delays may occur if:
- Documentation is incomplete
- Additional information is required
Submitting a complete and accurate application helps ensure faster approval.
Ready to Simplify Your MHRA Medical Device Registration?
Why Choose Operon Strategist for MHRA Medical Device Registration?
Operon Strategist provides end-to-end support for MHRA medical device registration, helping manufacturers achieve faster and smoother approvals.
Our services include:
- Device classification and regulatory strategy
- UK Responsible Person (UKRP) support
- Technical documentation preparation
- UKCA marking compliance
- Complete registration support
We also assist with ISO 13485, CE Marking, FDA 510(k), and global regulatory approvals, ensuring a seamless compliance journey.
FAQs
What is MHRA medical device registration?
MHRA medical device registration is the mandatory process of registering medical devices and IVDs with the UK regulatory authority before they can be legally sold in Great Britain.
Is MHRA registration required for all medical devices?
Yes, all medical devices and IVDs must be registered with MHRA before being placed on the UK market.
Who needs a UK Responsible Person (UKRP)?
Non-UK manufacturers must appoint a UK Responsible Person (UKRP) to handle device registration and communication with the MHRA.
What is the cost of MHRA medical device registration?
The MHRA charges a standard fee of £240 per registration, which can cover multiple devices if they are grouped correctly.
How long does MHRA registration take?
The MHRA typically reviews applications within 5 working days, although timelines may extend if additional information is required.