During COVID-19, there was lots of disruption in the world, which changed how things used to work. Due to travel restrictions and lockdowns to stop the virus from spreading, people shifted their work to online from offline mode. Information technology use reached its high time usage due to its high demand, as this was the only way to remain connected to other people. Due to corona, the regulatory bodies were too unable to perform their periodic audits and operations, which resulted in the halting of the manufacturing process, and supply chain and resulted in less availability of required products in the market. Traditionally medical device auditing is done on-site at the premises of the manufacturer/supplier but during the global pandemic notified body decided to explore other alternatives so that auditing does not stop and the flow of devices in the market does not get disrupted. Thus, regulatory bodies too decided to move their operations online and started with the Hybrid audit. In December 2022, MDCG released a guidance called MDCG 2022-17, an MDCG position paper on hybrid audits. As a Quality Mangement consultant we know how notified bodies work and hence, we assist medical device manufacturers in implementing effective Quality Management Services.
Hybrid Audit
A “Hybrid Audit” can be defined as an audit that takes place on the premises of the manufacturer/ supplier or the sub-contractor where at least one auditor is present on the premises, and the other team members of the audit are participating from other places using different technologies.
Hybrid Audit Vs Remote Audit
As per the British Standard Institute (BSI), in the Hybrid audit, one auditor must be present on the premises of the audit site, using technology to conduct the audit successfully. The remote audit is defined as the entire audit process being conducted online with the help of technology. The auditor can execute a remote audit from the manufacturer’s premises.
As Per the MDCG Paper on Hybrid Audits:
EU-MDR, which came into effect, is responsible for regulating the sale, quality, and distribution of medical devices. Any medical device being marketed must comply with the EU-MDR regulations; EU bodies were concerned that if notified bodies did not conduct conformity assessments on time, then it would disrupt the market flow of products. Thus, MDCG advised for hybrid audit to conduct medical device auditing in a timely and effective manner, and products will remain to comply with the regulations.
MDCG agreed on the term Hybrid it decided on the following definitions:
- A ‘hybrid audit’ should be understood as an audit on the premises of the manufacturer or its supplier(s) and/or subcontractor(s) where at least one auditor is present on the premises and other members of the audit team are participating from different locations using information and communication technologies (ICT).
- During the Hybrid audit the auditor (s) on the premises of the auditee of the other audit team members can be present either from the opening meeting to the closing meeting just for part of that time.
- The conformity assessment activity during the hybrid audit must be included in the audit plan that can be carried out either on the premises of the auditee, from elsewhere, or simultaneously from the auditee’s premises and elsewhere. The auditee should be involved in any case.
- The Notified bodies needs to make sure while establishing their hybrid audit plan that they plan sufficient time to audit the relevant processes on the auditee’s premises and identify and document those parts of the conformity assessment activities which were carried out on the auditee’s premises or using information technology.
Note: The MDCG may review the Hybrid audit position paper and determine whether modifications are needed based on the experience gained. The MDCG advises NBCG-Med to elaborate on operational elements, including identifying aspects to be audited on the auditee’s premises.
Benefits of Hybrid Audit
Since under no circumstances notified bodies are allowed to skip the medical device auditing hybrid audit seems to as a good option as it has its own benefits discussed below:
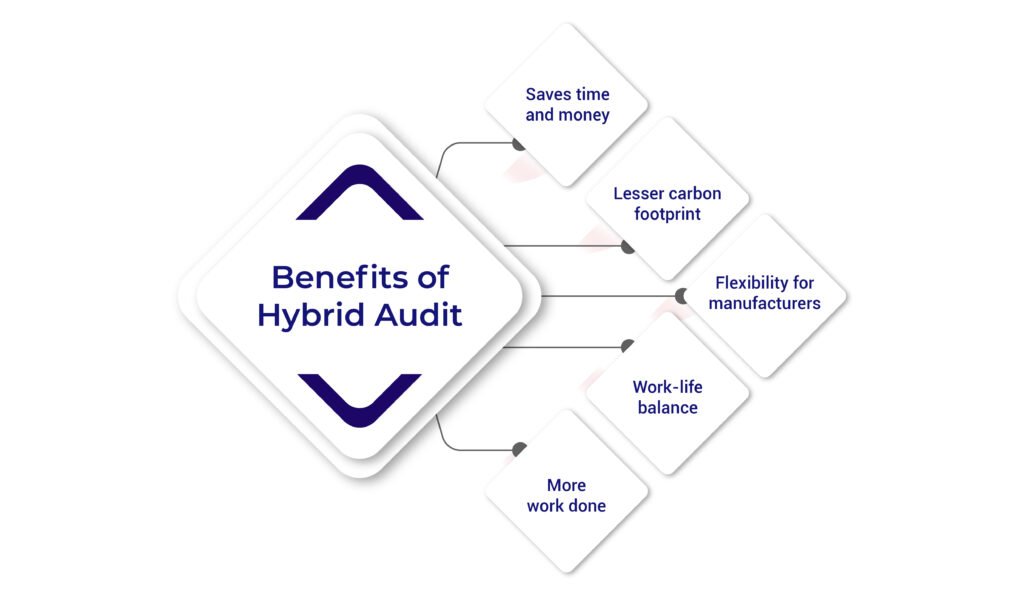
- Saves time and money Hybrid audit saves the auditor and auditee lots of time since they can work in half-virtual mode, leveraging both to plan their schedules, thus saving time and effort. Since only one auditor needs to travel, it will save money on traveling and staying.
- Lesser carbon footprint – Since the audit will involve less travel for the audit, the carbon footprint generated will be lesser, helping the environment too. Thus, one can conclude that Hybrid audits are environmentally friendly.
- Flexibility for manufacturers – Hybrid audits give enough time for manufacturers to plan their audits and provide them with the flexibility to conduct audits even if their time zones are different, allowing manufacturers to conduct them at their team’s convenience.
- Work-life balance – Since the team can be present on-premises or in a remote setting during hybrid audits, it will help them to have a better work-life balance. It will allow everyone to attend the audits even if they are dealing with something urgent and personal.
- More work done – Since one auditor can be present on the site premises and the other can be present in the remote setting, the person sitting in a remote setting can conduct more than one audit in a day, saving time and getting more work done in lesser time, implying hybrid audits can get more work done in lesser time.
The medical devices firms were anxious; as of Oct 2022, there were only 31 notified bodies. The corona has caused a lot of delays now the lack of notified bodies caused more disruption. To overcome these hurdles, EU-MDR came up with hybrid audits to ease the medical device firms and notified bodies of the burden. The MDCG has already released guidance where they have mentioned the clear meaning of Hybrid to avoid confusion. Unfortunately, there is nothing the manufacturers can do about the lack of notified bodies. Still, they can remain prepared for hybrid audits to avoid further marketing delays.




