CE Marking for Medical Devices: Process & EU MDR Requirements
What Is CE Marking for Medical Devices?
.CE marking (commonly referred to as the CE mark) is a mandatory certification that indicates a medical device complies with European Union (EU) regulatory requirements under the Medical Device Regulation (EU MDR 2017/745).It allows manufacturers to legally market and distribute their medical devices across all EU member states and European Economic Area (EEA) countries.
CE marking confirms that the device meets essential requirements related to:
- Safety
- Performance
- Quality
- Risk management
Without CE marking, medical devices cannot be sold in the European market.
CE Marking Requirements Under EU MDR
To obtain CE marking for medical devices, manufacturers must comply with strict regulatory requirements defined under EU MDR.
Key requirements include:
- Device Classification based on risk level
- Technical Documentation demonstrating compliance
- Clinical Evaluation proving safety and performance
- Risk Management as per ISO 14971
- Quality Management System (QMS) such as ISO 13485
- Post-Market Surveillance (PMS) and vigilance systems
Manufacturers are fully responsible for ensuring continuous compliance throughout the product lifecycle
Medical Device Classification in Europe
Medical devices in Europe are classified based on risk:
- Class I – Low risk (e.g., non-invasive devices)
- Class IIa – Medium risk
- Class IIb – Medium to high risk
- Class III – High risk (e.g., implantable devices)
Higher classification results in more stringent regulatory requirements and mandatory involvement of a notified body.
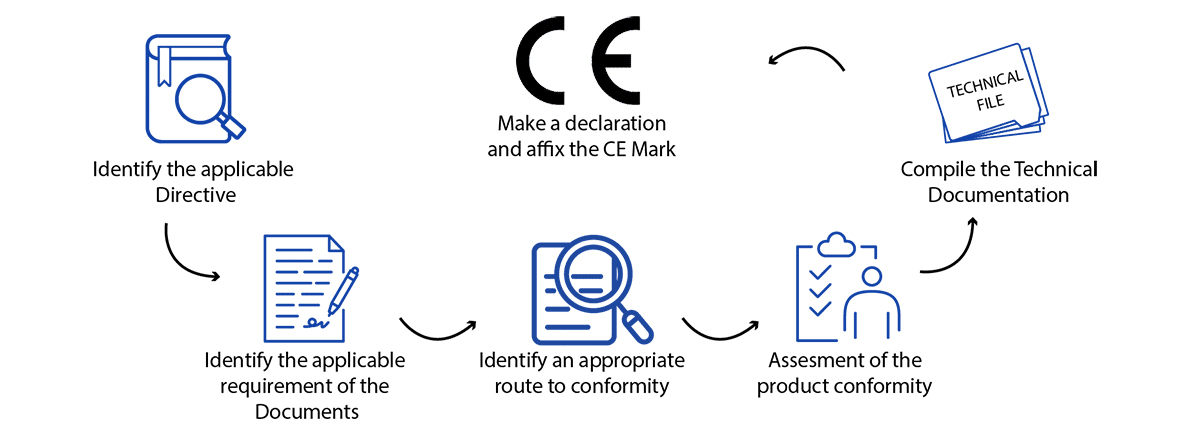
How to Get a CE Marking for a Medical Device?
While “CE” isn’t a quality mark, adherence to the EU Medical Devices Regulation (MDR 2017/745) necessitates meeting precise standards of performance, quality, safety, and efficacy tailored to your product type. Operon Strategist’s comprehensive guide outlines the current European CE approval process for medical devices. Nonetheless, the fundamental procedure for obtaining a CE mark for medical devices typically involves the following steps:
- Determine if your product aligns with the definition of a medical device as outlined in the EU MDR.
- Determine the medical device classification in Europe for your medical devices.
- Implement a Quality Management System (QMS) if it applies to your device. Many companies utilize ISO 13485 to fulfill these requirements.
- Prepare a CE Marking Technical File or a Design Dossier.
- Compile a Clinical Evaluation Report (CER) following the guidelines of MEDDEV 2.7/1 rev4 and the MDR.
- Select and designate a European Authorized Representative (EC REP) to act on your behalf within the EU if you lack a physical presence in Europe.
- Subject your QMS and Technical File/Design Dossier to an audit by a Notified Body, unless your device falls under Class I, is non-sterile, and lacks a measuring function.
- Obtain CE certificate for medical devices and ISO 13485 certificates from your designated Notified Body.
- Prepare a EU Declaration of Conformity (DoC) confirming that your device complies with the regulations outlined in the MDR.
If you need more clarity on obtaining CE certification for your medical devices, contact us on WhatsApp or call 918767980322
Declaration of Conformity
After ensuring that the safety and performance criteria have been satisfied and the requisite technical documentation is ready, the manufacturer is obligated to date, sign, and maintain a declaration of conformity. This declaration serves as proof that the product adheres to the standards outlined in the executive order.
Recommended blog: EU Declaration of Conformity for Medical Devices
Quality Management Systems - Requirements for Regulatory Purposes.
- ISO 13485:2016 – The ISO 13485:2016 standard lays out the rules for how to manage the quality of medical devices. Most medical device makers follow this standard because it’s a good way to make sure they meet the quality management system (QMS) requirements in the MDR.
- FDA 21 CFR Part 820 – FDA 21 CFR Part 820 sets the quality system rules for medical device manufacturers. This regulation is the current standard for quality management systems for medical devices used in the United States.
CE Marking Cost and Effort Factors
The cost of CE marking for medical devices is not fixed and depends on multiple regulatory and technical factors.
Key influencing factors include:
- Device classification and risk level
- Product complexity and intended use
- Testing and clinical evaluation requirements
- Technical documentation scope
- Notified body involvement
Due to these variables, CE marking is evaluated on a case-by-case basis.
CE Marking Timeline
The timeline for CE marking varies depending on device complexity and regulatory pathway.
- Lower-risk devices typically follow a shorter approval process
- Higher-risk devices require more time due to clinical evaluation and regulatory review
Early planning and proper documentation can significantly reduce approval timelines.
Get Your Tailored CE Marking Roadmap with Clear Timelines and Expert Guidance
European Authorized Representative (EC REP)
Manufacturers outside the EU must appoint a European Authorized Representative (EC REP) to place devices in the European market.
The EC REP:
- Acts as a regulatory contact point
- Maintains technical documentation
- Communicates with EU authorities
- Supports compliance throughout the lifecycle
What Are Notified Bodies?
Notified Bodies are independent organizations designated by EU member states to assess medical device conformity.
They:
- Conduct audits and assessments
- Review technical documentation
- Issue CE certificates
- Perform ongoing surveillance
Notified body involvement is mandatory for Class IIa, IIb, and III devices.
Streamline EU MDR Compliance With Operon Strategist
To simplify the approval process, Operon Strategist, a leading EU MDR consultant, supports manufacturers in the following:
- Assisting with medical device classification
- Verifying applicable standards and testing requirements
- Compiling Technical Files or reviewing existing documentation
- Reviewing marketing materials, labels, and user manuals for compliance and consistency
- Ensuring adherence to Essential Requirements
- Crafting Clinical Evaluation Reports based on provided clinical data
- Implementing, adjusting, and maintaining a quality system (usually ISO 13485) to meet European and international criteria
- Providing European Authorized Representative services
- Conducting risk assessment and management following ISO 14971
- Developing vigilance and post-market surveillance procedures
MDR CE marking Approval process will vary according to the Class of medical device, as per the EU MDR 2017/745, CE marking Approval consist of a few more things such as product quality, technical dossier submission to Notified Body, clinical evaluation, and so on.
Contact us to learn how we can assist you in obtaining CE marking for your medical device in Europe.
Get Your Medical Device CE Marked Fast and Easy With Operon Strategist!
FAQS
1. Check the CE mark’s placement and appearance
2. Verify the identification number of the Notified Body
3. Check the product’s documentation
4. Check the manufacturer’s address
In medical terms, “CE” stands for “Conformité Européene,” which translates to “European Conformity” in French. All medical devices and IVDs, with the exception of custom-made devices and those for clinical investigations, are required to display the CE mark.
Yes, medical devices generally need to be CE marked. The CE marking indicates that a medical device conforms to the essential safety and performance requirements outlined by European Union (EU) regulations. However, there are some exceptions, such as custom-made devices and those intended for clinical investigations, which may not require CE marking.
Typically, under the current framework, CE certificates issued by Notified Bodies remain valid for about three years. However, for certain high-risk devices, this validity period might be reduced to one year. It’s important to note that the continuity of your CE certification relies on upholding your quality system certification.
The duration to acquire a CE mark for a medical device typically spans four to six weeks on average. However, the timeline can vary, influenced by factors such as test outcomes, necessary product adjustments, and the promptness of providing technical documentation. It’s important to note that the specific CE marking process varies for each product.
For Class 1 medical devices, obtaining CE marking can be accomplished through self-declaration according to the MDR. This means that neither Notified Body certification nor approvals from certification bodies are necessary. Class 1 devices are considered to have minimal risk, allowing manufacturers to self-certify them.
Simplify Your CE Mark Journey
Get expert guidance on CE marking for medical devices. From classification to clinical evaluations, technical documentation, and EC REP services, we handle it all. Ensure compliance with EU MDR seamlessly.