CDSCO Registration for Medical Devices in India
Get your CDSCO import or manufacturing license approved without delays, rejections, or compliance risks.
Whether you are an importer, manufacturer, or startup, we help you get end to end regulatory approval under the Medical Device Rules 2017.
✔ Import License (MD-15)
✔ Manufacturing License (Class A to D)
✔ End to End Documentation & Approval Support
What is CDSCO Registration?
CDSCO registration is a mandatory approval required to import, manufacture, or sell medical devices in India.
It is regulated by the Central Drugs Standard Control Organization under the Medical Device Rules 2017.
If your product falls under notified medical devices, you must obtain CDSCO approval before entering the Indian market.
TYPES OF CDSCO LICENSES IN INDIA
CDSCO Import License (MD-15)
A CDSCO Import License is required if you plan to bring medical devices into India for sale or distribution.
When you need it:
If your product is manufactured outside India and you want to sell it in the Indian market.
Who should apply:
- Foreign manufacturers
- Indian importers and distributors
- Authorized agents
Key details:
- Application: MD-14
- License issued: MD-15
- Validity: 5 years
- Requires Indian Authorized Agent
CDSCO Manufacturing License
A CDSCO Manufacturing License is required to manufacture or assemble medical devices within India.
When you need it:
If you are producing or assembling medical devices in India for sale or export.
Who should apply:
- Indian manufacturers
- Startups setting up manufacturing units
- Companies doing assembly or packaging
Key details:
- Class A & B: State Licensing Authority
- Class C & D: Central Licensing Authority
- Requires GMP compliance
- Based on device classification
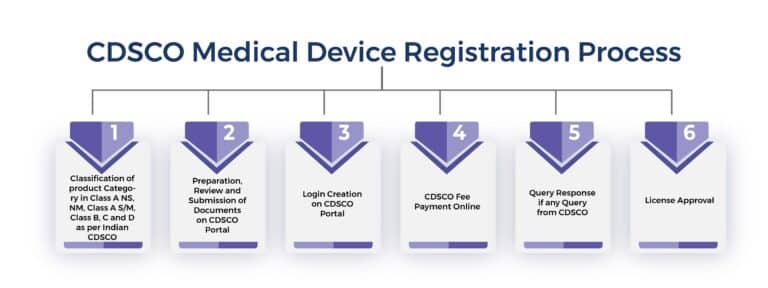
CDSCO REGISTRATION PROCESS IN INDIA
Here is the step by step process:
Step 1: Device Classification
Identify whether your device falls under Class A, B, C, or D.
Step 2: Documentation Preparation
Prepare technical documents like DMF, PMF, ISO certificates, and labeling.
Step 3: Application Filing
Submit your application on the CDSCO online portal.
Step 4: Review & Query Handling
Regulators may raise queries that must be addressed properly.
Step 5: License Approval
Once approved, you receive your CDSCO license.
Read in Detail: Medical Device Registration process in India
DOCUMENTS REQUIRED FOR CDSCO REGISTRATION
- Device Master File (DMF)
- Plant Master File (PMF)
- ISO 13485 Certificate
- Free Sale Certificate
- Technical File
- Labeling and packaging details
- Authorized Agent agreement (for importers)
From Registration to Market Access: Operon Strategist Paves the Way for Your Medical Devices in India
Import vs Manufacturing License (Quick Comparison)
| Criteria | Import License | Manufacturing License |
|---|---|---|
| Purpose | Import devices into India | Manufacture devices in India |
| Applicant | Importer / Authorized Agent | Manufacturer |
| Forms | MD-14 → MD-15 | MD-3, MD-5, MD-7, MD-9 |
| Authority | CDSCO | State + CDSCO |
| Use Case | Foreign products | Local production |
COMMON CHALLENGES
Most companies face delays due to:
- Incorrect device classification
- Incomplete documentation
- CDSCO query handling issues
- Lack of regulatory clarity
- Rejections due to non compliance
This leads to months of delay and lost market opportunity.
Many companies in India bring raw materials, semi-finished products, or components to India. In order to sell this finished medical device, the final assembly and final packaging of the medical device in India require a CDSCO manufacturing license. However, if you require a complete finished product outside India, you will require a CDSCO import license.
CDSCO Forms for Registration of Existing Medical Devices
| Applicant | Risk/Class | Type of License | Forms |
|---|---|---|---|
| Importer | A,B,C,D | Importer License | Application: MD-14 Permission: MD-15 |
| Manufacturer | A,B | Manufacturing License | Application: MD-3 Permission: MD-5 |
| Loan License | Application: MD-4 Permission: MD-6 | ||
| C,D | Manufacturing License | Application: MD-7 Permission: MD-9 | |
| Loan License | Application: MD-8 Permission: MD-10 |

Latest CDSCO Update
- All medical devices now require mandatory registration for import and sale in India, effective October 1, 2023.
- Only exemption: Only Class C and Class D devices that were registered before October 1, 2023, are eligible for the next 6 months.
Know more: 6 Month Extension for CDSCO Class C & Class D Non-Notified Medical Device
Classification of Medical Devices as per Indian CDSCO Regulations
Medical devices in India are classified based on the level of risk they pose to patients and users, as defined under the Medical Device Rules 2017 by the Central Drugs Standard Control Organization.
The classification determines the type of approval required, the licensing authority, and the level of regulatory scrutiny.
Medical devices are divided into four categories:
Here are the four categories of medical devices as per CDSCO:
- Class A:- Low-risk medical devices such as stethoscopes, bandages, and other basic medical instruments that have minimal or no potential to harm patients or users.
- Class B:- Low-to-moderate-risk medical devices such as blood pressure monitors, syringes, and needles that may cause harm to patients or users if they malfunction, but the harm is not life-threatening.
- Class C:- Moderate-to-high-risk medical devices such as artificial heart valves, orthopedic implants, and catheters that have the potential to cause serious harm or injury to patients if they malfunction.
- Class D:- High-risk medical devices such as pacemakers, heart-lung machines, and ventilators that are critical to the health and survival of patients and could cause serious harm or death if they malfunction.
What are the Benefits of CDSCO Registration?
CDSCO registration is a crucial step for any medical device manufacturer/ Importer looking to enter the Indian market. It provides several benefits, including:
- Market access: CDSCO registration allows the manufacturer to sell and distribute their products in the Indian market, which is one of the largest pharmaceutical and medical device markets in the world.
- Brand recognition: CDSCO registration enhances the manufacturer’s brand recognition and credibility, as it demonstrates that its products have met rigorous safety, efficacy, and quality standards.
- Regulatory compliance: CDSCO registration ensures that the manufacturer is compliant with Indian regulatory requirements, including GMP and MDR.
- Competitive advantage: CDSCO registration provides a competitive advantage, as it is a prerequisite for participation in tenders and procurement by government and private institutions.
Experience a Smooth Path to Device Registration—Let Our Experts Drive Your Product to Market Success!
*Note- The Bureau of Indian Standards (BIS) certification is required in India for medical devices to ensure their adherence to specific quality, safety, and performance standards mandated by the Indian government. The BIS certification is a mark of quality assurance and compliance with set standards.
Manufacturers of products that would potentially require in-country testing to BIS standards should consider seeking registration as soon as possible. Additionally, all Class A and B products are required to have an Import License. Read more about BIS Certification for Medical Devices.
How Operon Strategist will Help you to Get a CDSCO License for Medical Devices Registration in India?
Operon Strategist is a medical device regulatory consultant and assists in CDSCO registration, We make sure that clients from various countries get hassle-free results within the timeframe.
Operon Strategist Step-by-step Approach for CDSCO Medical Devices Registration in India
- Regulatory Guidance: Operon Strategist provide expert knowledge on the regulatory requirements set by the CDSCO, for medical device registration in India. We help clients understand the complex regulatory landscape.
- Classification and Compliance: Help to classify medical devices based on risk and purpose, ensuring that they comply with CDSCO guidelines. Different classes of devices have different requirements, and we help navigate this process.
- Documentation Preparation: Assist in preparing and organizing the necessary documentation required for CDSCO registration. This includes compiling technical files, quality management system documents, and clinical trial data.
- Online Submission: Operon Strategist guide clients through the online submission process, helping them submit their applications to the CDSCO portal.
- Review and Audit: Review the documents and applications to ensure they meet CDSCO standards. We may conduct pre-audits to verify compliance with regulations and provide guidance on rectifying any issues.
- Communication with CDSCO: Operon act as intermediaries between clients and the CDSCO, handling queries and requests for additional information from the regulatory authority.
- Market Strategy: Provide insights into the Indian medical device market, helping to develop market entry and expansion strategies.
- Post-Approval Support: Including renewals, variations, and other ongoing regulatory requirements.
- Quality Management System: Offer a guidance on establishing or improving a quality management system to meet CDSCO requirements.
As a CDSCO registration consultant, Operon Strategist facilitate the entire process of gaining regulatory approval for medical devices in India. Our expertise streamlines the process, increases the chances of successful registration, and ensures that your medical device products meet safety, efficacy, and quality standards. For the details of CDSCO registration, you can Contact Us or WhatsApp us at +91 9403892834.
Visit Our Manufacturing Blogs for More Information.
FAQs
How to register a medical device with CDSCO?
Operon strategist will assist you to register your medical device with CDSCO. Registration of medical devices with CDSCO is an online process.The CDSCO registration process is tedious and it requires medical device regulatory expertise to get the license. Operon Strategists help in preparation of documents,review of documents by experts , submission of documents , guidance on online payment to CDSCO and resolving queries if any . Operon Strategist also assists to conduct pre- audits to verify compliances and also provide audit assistance in manufacturing license
What is voluntary registration in CDSCO?
Voluntary registration in the temporary registration process for all non-notified Class C and Class D products as per CDSCO notification. This is an online registration process and there is no CDSCO fee applicable for voluntary registration.
Who approves medical devices in India?
The Drug Controller General of India (DCGI) within the Central Drugs Standard Control Organization (CDSCO), which is part of the Ministry of Health and Family Welfare, regulates medical devices and IVDs.