
Top 5 Businesses to Invest in 2025
As global markets evolve and new technologies reshape industries, 2025 presents a fresh landscape of opportunities for savvy investors and

As global markets evolve and new technologies reshape industries, 2025 presents a fresh landscape of opportunities for savvy investors and

The European Union Medical Device Regulation (EU MDR) indeed represents a substantial shift in the regulatory landscape for medical device
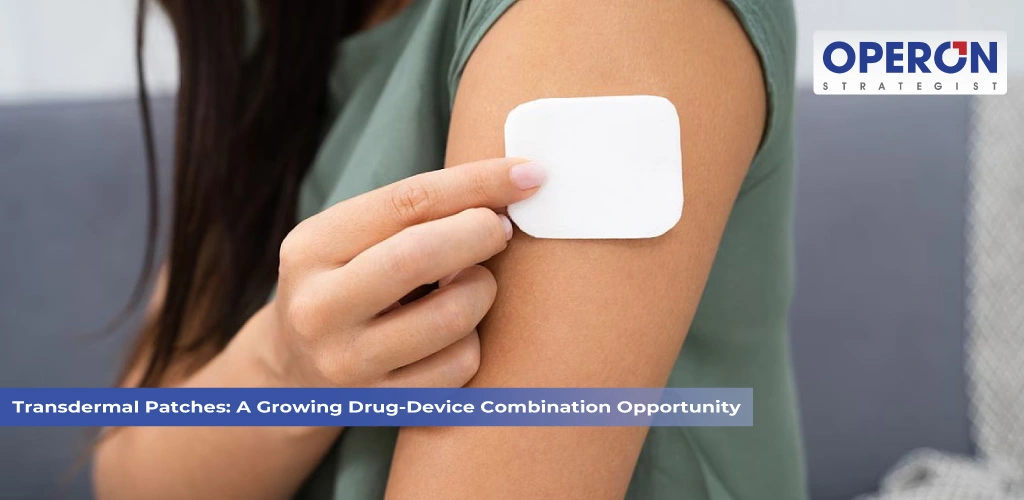
Introduction Transdermal patches have emerged as one of the most innovative drug delivery systems in modern healthcare. These patches deliver

Introduction When navigating the complex regulatory landscape of medical device manufacturing in the European Union, one term you’ll frequently encounter
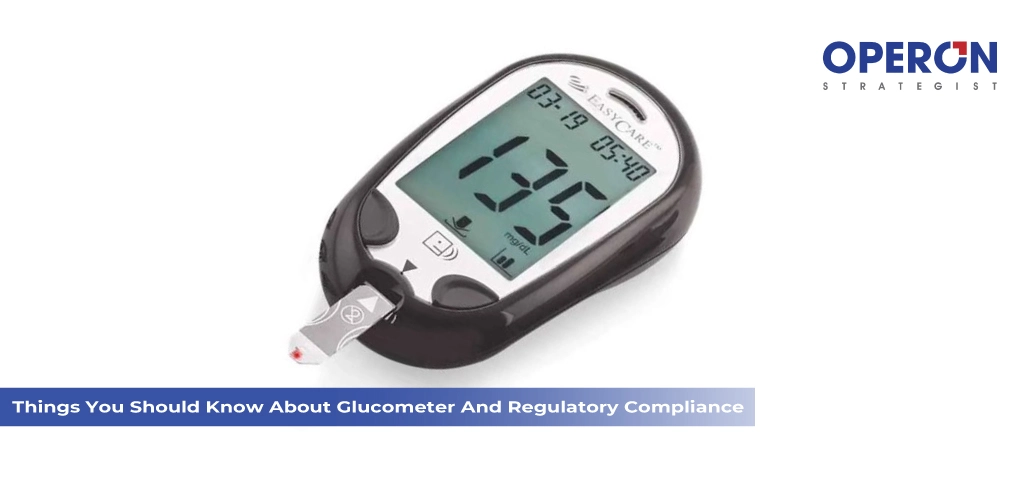
What are Glucometer Devices? A glucometer, also known as a blood glucose meter, is a portable medical device used to

Introduction: In the realm of healthcare, innovation often emerges at the intersection of different disciplines. One such area is the

The requirements for MDR classification for medical devices are nearly identical to those in the current Medical Devices Directive (MDD).

Overview The medical device industry faces unique challenges when it comes to custom clearance due to stringent regulations and administrative

Overview Rehabilitation medical devices play a pivotal role in restoring independence and improving the quality of life for individuals with

2025 Medical Device Trends The global medical device industry is stepping into 2025 with unprecedented momentum, driven by cutting-edge technologies,